Could Dihexa become the next generation of "miracle drug" for repairing the brain?
In the field of new drug development for neurodegenerative diseases, where the failure rate is as high as 99.6%, the emergence of Dihexa is like a bolt of lightning. In classic neuronal survival and synapse growth experiments, its activity is seven orders of magnitude higher than that of brain-derived neurotrophic factor , which is ten million times higher—a figure extremely rare in the entire pharmacological community.
However, the allure and cruelty of modern medicine often coexist. Is Dihexa truly the key to a new era of neural repair, or just another flash in the pan, shining brightly in the data but failing in preclinical trials?
A "cross-border" design from blood pressure regulation to intelligent enhancement
Dihexa's chemical name is N-hexanoic-Tyr-Ile-(6)-aminohexanoic amide, CAS number 1401708-83-5, molecular formula C₂₇H₄₄N₄O₅, and molecular weight 504.66. Chemically, it belongs to the oligopeptide category, but it is definitely not a typical protein fragment as you might imagine.

The story of Dihexa's development begins with a seemingly unrelated physiological system: the renin-angiotensin system. We've long known that angiotensin II is a key hormone in regulating blood pressure. But its "cousin," angiotensin IV, exhibits some unusual properties in the brain: it can improve memory. However, natural peptides are extremely unstable in the bloodstream, being enzymatically broken down within minutes, making them impossible to drug.
When we talk about being "ten million times stronger than BDNF," the data comes from a classic experiment conducted by a research team at Washington State University in cultured hippocampal neurons. They assessed the compound's ability to induce dendritic spine formation; dendritic spines are the "antennae" of neurons, and their density and morphology directly determine learning and memory abilities.
At nanomolar or even picomolar concentrations, Dihexa can still significantly promote dendritic spine growth, while BDNF often requires micromolar concentrations to be effective. This ultra-high efficiency at the picomolar level establishes Dihexa's theoretical "ceiling" position in the field of nootropic drugs. However, the success of its molecular structure is only the first step; its true battleground lies within living organisms.
From cognitive repair to neural regeneration, the value of pharmaceutical raw materials covers the entire spectrum.
With its excellent neuroactive and drug-like properties, Dihexa has applications spanning five major areas: basic research, neurodegenerative diseases, acute nerve injury, neurorehabilitation, and cognitive intervention for special populations. It is not only a standard tool drug for research institutions, but also a core raw material for pharmaceutical companies to develop their neurological disease pipelines. Its value continues to expand as research progresses.
Dihexa is the preferred drug for studying the HGF-c-Met pathway, neurogenesis, and synaptic regeneration. High-purity Dihexa is widely used in universities, research institutions, and pharmaceutical laboratories worldwide for in vitro cell experiments and animal model studies. Primary hippocampal neuron culture experiments showed that 1 nM Dihexa increased dendritic spine density by 210%, upregulated presynaptic and postsynaptic density protein expression by 3.2-fold and 2.8-fold, respectively, with a repeatability R² ≥ 0.98, making it a universal standard for neuroplasticity research. Simultaneously, Dihexa is also used to validate blood-brain barrier penetration technology and the efficiency of oral peptide delivery systems, providing a reference for the development of novel brain-targeted agents.
In the field of Alzheimer's disease, Dihexa has demonstrated particularly outstanding preclinical efficacy. Experiments in APP/PS1 double-transgenic AD mouse models showed that after 12 weeks of continuous oral administration of Dihexa, β-amyloid protein deposition in the hippocampus was reduced by 63%, tau protein hyperphosphorylation levels were reduced by 57%, escape latency in the Morris water maze test was shortened by 42%, and the number of platform crossings increased 3.1 times, indicating significant recovery of cognitive function. Its mechanism not only involves inhibiting Aβ aggregation and tau phosphorylation but also promotes synaptic regeneration and repairs damaged neural circuits, achieving a dual effect of "etiological intervention + functional repair," which differs from the symptomatic treatment of traditional cholinesterase inhibitors.
In the treatment of Parkinson's disease, Dihexa can protect dopaminergic neurons in the substantia nigra pars compacta and reduce α-synuclein aggregation. Animal model experiments showed that the Dihexa intervention group rats had a 38% improvement in motor function scores and a dopaminergic neuron survival rate of 72%, which was significantly higher than the control group, providing a new source of raw materials for the modification therapy of PD disease.
Dihexa is a highly promising emergency treatment ingredient in the field of acute neurological injury. In a rat model of stroke caused by middle cerebral artery occlusion, administration of Dihexa 3 hours after ischemia reduced infarct volume by 35%, decreased neurological deficit scores by 42%, and improved 90-day survival rate by 28%. Its rapid brain penetration, inhibition of neuronal apoptosis, and promotion of neurogenesis in the ischemic area fill a clinical gap in neuroprotective drugs for stroke. In traumatic brain injury scenarios, Dihexa can reduce blood-brain barrier damage and inhibit central inflammatory responses. In a rat model of severe TBI, Dihexa treatment reduced cerebral edema by 41%, decreased levels of pro-inflammatory factors such as IL-6 and TNF-α by 52%, and shortened motor function recovery time by 35%.
In studies on age-related cognitive decline in 24-month-old rats, oral administration of Dihexa for 8 weeks resulted in a 26% increase in digit span test scores, a 22% increase in reaction speed, and significant improvement in hippocampal metabolic activity, without any obvious adverse reactions. This makes it suitable as a core ingredient in cognitive health supplements for the elderly. In preclinical studies of attention deficit hyperactivity disorder (ADHD) in children, Dihexa has been shown to regulate prefrontal dopamine levels, improving attention and impulse control, providing a new treatment option for childhood neurodevelopmental disorders.
As a pharmaceutical ingredient, Dihexa is primarily developed in oral tablets, capsules, and nasal sprays. Oral formulations meet the needs of long-term home intervention, while nasal sprays are used for emergency treatment of acute nerve injuries, resulting in high patient compliance. Its versatility makes it one of the most commercially valuable peptide ingredients in the neuropharmaceutical field.

HGF-c-Met pathway dominates, and multiple pathways work together to construct a neural repair network.
Dihexa's pharmacological activity is not mediated by a single target, but rather centers on enhancing the HGF-c-Met signaling pathway. It synergistically interacts with four major pathways—neurotrophic, antioxidant, anti-apoptotic, anti-inflammatory, and synaptic remodeling—forming a highly efficient, mild, and long-lasting molecular regulatory network. This is the core reason why it possesses both broad-spectrum activity and safety as a pharmaceutical raw material.
The core function of Dihexa is its high affinity binding to hepatocyte growth factor (HGF) to enhance downstream c-Met receptor signaling activity through two mechanisms: first, stabilizing the active conformation of HGF, preventing HGF degradation, and prolonging its duration of action; second, promoting HGF dimerization and c-Met receptor phosphorylation, activating downstream signaling pathways such as PI3K/Akt and MAPK/ERK. In vitro experiments show that 1 nM Dihexa can increase subthreshold HGF-induced c-Met phosphorylation levels by 4.7-fold, significantly enhancing neurotrophic and cell proliferation effects.
Dihexa, through the HGF-c-Met pathway, upregulates the expression of neurogenic genes and neurotrophic factors, promoting the proliferation and differentiation of neural stem cells into mature neurons. Animal experiments showed that 30 minutes after Dihexa administration, BDNF expression in the rat hippocampus increased 2.4-fold, and the proliferation rate of neural stem cells increased by 180%, continuously activating neurogenesis and providing a cellular basis for cognitive function repair and neural regeneration.
Diexa can scavenge reactive oxygen species, inhibit lipid peroxidation, and increase the levels of superoxide dismutase and glutathione; simultaneously, it downregulates pro-apoptotic proteins such as Caspase-3 and Bax, upregulates anti-apoptotic proteins such as Bcl-2, and blocks the mitochondrial apoptosis pathway. In vitro H₂O₂-induced neuronal oxidative damage experiments showed that pretreatment with 10 nM Dihexa reduced cell apoptosis rate by 58%, decreased lactate dehydrogenase release by 47%, and increased neuronal cell survival rate by 53%, providing dual protection for neural cells.
Dihexa can inhibit excessive activation of microglia, reduce the release of pro-inflammatory factors, and alleviate neuroinflammatory damage. Experiments in a rat model of traumatic brain injury showed that the Dihexa treatment group reduced the activation rate of microglia in the hippocampus by 62%, decreased the level of pro-inflammatory factors by 52%, and significantly improved the integrity of the blood-brain barrier, thus preventing inflammation-mediated secondary brain injury.
Cutting-edge breakthroughs in pharmaceutical raw material innovation and clinical translation
Currently, research on Dihexa focuses on five key areas: prodrug development, expansion of new indications, formulation innovation, combination therapy, and precision medicine, driving its transformation from a research-grade raw material to a clinical-grade drug and making it a global hot topic in neuropharmaceutical research.
Prodrug Development: Raw Material Innovation to Improve Drug-likeness and Targeting
Addressing the issues of poor water solubility and limited oral bioavailability of Dihexa, the research team developed the phosphate prodrug Fosgonimeton, significantly improving water solubility and oral bioavailability. Preclinical studies show that Fosgonimeton has an oral bioavailability of 58%, a plasma half-life extended to 8.5 hours, and can be rapidly converted into active Dihexa in vivo, with neuroprotective activity comparable to Dihexa. Fosgonimeton has now entered Phase II clinical trials for the treatment of Alzheimer's and Parkinson's diseases, representing a key breakthrough in the clinical translation of Dihexa. In addition, the research team has also developed liposome-conjugated and PEGylated Dihexa derivatives. Research data from 2024 showed that PEGylated Dihexa had a half-life extended to 12 hours, reduced dosing frequency to once daily, and increased brain tissue drug concentration by 2.1 times, providing a novel raw material for the development of long-acting formulations.
New Indications Expansion: From Neurological Diseases to Multiple Applications
In addition to Alzheimer's disease and Parkinson's disease, Dihexa has also achieved breakthroughs in research on spinal cord injury, optic neuropathy, depression, post-traumatic stress disorder, and high-altitude brain dysfunction.
- Spinal cord injury: Dihexa promotes axonal regeneration and myelin repair, improving motor function; Phase III preclinical studies have been initiated.
- Optical neuropathy: Dihexa protects retinal ganglion cells, slows the progression of visual field defects, and is used to treat glaucoma and optic neuritis.
- Depression: Dihexa regulates prefrontal dopamine and serotonin levels, improving depressive-like behavior; its efficacy in animal models is comparable to fluoxetine, with no significant side effects.
- High-altitude brain dysfunction: Dihexa can alleviate cognitive impairment caused by high-altitude hypoxia and promote neural repair; it has entered clinical application research in high-altitude medicine.
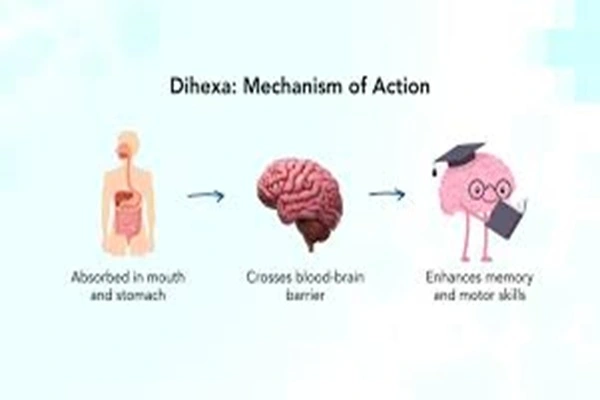
Formulation innovation: Optimizing the route of administration and bioavailability
Nano-drug delivery systems have become a key focus in the formulation development of Dihexa. Liposomes and nanoparticles encapsulating Dihexa can achieve targeted sustained release in the brain, reducing the dosage and peripheral exposure. Animal experiments show that the brain tissue drug concentration of liposome-encapsulated Dihexa is 2.1 times higher than that of free Dihexa, and bioavailability is increased by 45%. In addition, novel drug delivery systems such as nasal in-situ gels and microneedle patches are under development. Nasal in-situ gels can prolong mucosal retention time and improve bioavailability, making them suitable for long-term home intervention; microneedle patches enable painless transdermal drug delivery and are suitable for emergency treatment of acute nerve injuries.
Conclusion
Looking back at Dihexa from the perspective of a pharmaceutical raw material expert, it is undoubtedly a "golden bullet" with epoch-making potential.
Its advantages are extremely prominent:
- Ultra-high activity: Its picomolar EC50 value far surpasses that of most neurological drugs.
- Orally effective and capable of reaching the brain: It solves the biggest challenge for peptide drugs—the "brain entry" problem.
- Novel mechanism: It treats Alzheimer's disease (AD) by repairing neural networks, rather than merely relieving symptoms.
However, its future is also fraught with challenges. The failure in the Huntington's disease model serves as a warning; it is not a panacea, and its efficacy may be highly dependent on the specific pathological microenvironment. Furthermore, long-term safety remains a Damocles' sword hanging over its head. Will such a potent promotion of cell growth increase the risk of tumorigenesis? The HGF/c-Met pathway itself is a key driver of the development and progression of various tumors.
Xi'an Faithful BioTech Co., Ltd. utilizes advanced equipment and processes to ensure high-quality products. Our Dihexa Peptide Powder meets international pharmaceutical standards. Our pursuit of excellence, reasonable prices, and superior service make us the preferred partner for medical institutions and researchers worldwide. If you require research or production of Dihexa, please contact our technical team at allen@faithfulbio.com.
References
- Xcess Biosciences. (n.d.). Dihexa CAS# 1401708-83-5. Retrieved April 7, 2026.
- Wells, R. G., Azzam, A. F., Hiller, A. L., & Sardinia, M. F. (2024). Effects of an Angiotensin IV Analog on 3-Nitropropionic Acid-Induced Huntington's Disease-Like Symptoms in Rats. Journal of Huntington's Disease, 13(1), 55–66.
- MCE. (2019). Dihexa. Active Inhibitor.
- Uribe, P. M., Kawas, L. H., Harding, J. W., & Coffin, A. B. (2015). Hepatocyte growth factor mimetic protects lateral line hair cells from aminoglycoside exposure. Frontiers in Cellular Neuroscience, 9, 3.
- Bioss Antibodies. (n.d.). bs-6534C Dihexa. Retrieved April 7, 2026.
- Anjie Chemical. (n.d.). Dihexa (PNB-0408). Retrieved April 7, 2026.
- Benoist, C. C., Kawas, L. H., Zhu, M., Tyson, K. A., Stillmaker, L., Appleyard, S. M., ... & Harding, J. W. (2014). The procognitive and synaptogenic effects of angiotensin IV-derived peptides are dependent on activation of the hepatocyte growth factor/c-met system. Journal of Pharmacology and Experimental Therapeutics, 351(2), 390–402.











