How can Forskolin Powder, a natural cAMP activator, become a core active pharmaceutical ingredient for multiple systemic diseases due to its unique diterpenoid structure?
Forskolin is a diterpenoid-type natural active ingredient isolated and extracted from the rhizome of Forsythia suspensa, a herbaceous plant in the Lamiaceae family. It is currently the only naturally occurring compound proven to directly and non-receptor-dependently activate adenylate cyclase. As a classic star molecule in the pharmaceutical field, high-purity Forskolin Powder, with its core advantages of "potently increasing intracellular cAMP levels, regulating physiological functions through multiple pathways, broad-spectrum pharmacological activity, and low toxicity," overcomes the limitations of traditional synthetic drugs, such as "single target, significant side effects, and easy drug resistance." It has become a core active pharmaceutical ingredient for drug development and clinical application in multiple fields, including ophthalmology, cardiovascular, metabolic, neurological, and oncology. From FDA-approved glaucoma eye drops to potential treatments for heart failure, obesity, and asthma, and further to cutting-edge research in neuroprotection, anti-tumor, and antiviral applications, Forskolin, through its unique structure-activity relationship, constructs a "cAMP-PKA-Epac" core signaling regulatory network, achieving precise regulation of cell metabolism, proliferation, differentiation, ion channels, and gene expression.
Precise structure-activity framework and physicochemical properties of hemispheric diterpenes
Forskolin, chemically named (3R,4aR,5S,6S,6aS,10S,10aR,10bS)-5-acetoxy-3-vinyldodecyl-6,10,10b-trihydroxy-3,4a,7,7,10a-pentamethyl-1H-naphtho[2,1-b]pyran-1-one, with the molecular formula C₂₂H₃₄O₇ and a precise molecular weight of 410.50 Da, is a typical tricyclic diterpenoid compound. Its molecular structure exhibits a highly specific combination of "decahydronaphthyl core + tetrahydropyran ring + polyhydroxy substitution + acetoxy group + vinyl side chain + epoxy bridging bond". This unique polycyclic, multifunctional structure endows it with potent adenylate cyclase activation activity, high lipophilicity, strong cell membrane permeability, and high target selectivity, which is the core structure distinguishing it from other diterpenoid compounds. Pharmaceutical-grade Forskolin Powder is a white to off-white crystalline powder with stable crystal form and controllable impurities, fully complying with USP, EP, and ICH Q7 pharmaceutical standards.

Forskolin's molecular structure consists of five key modules, each corresponding to a specific biological activity. The structure-activity relationship is highly defined; any modification to any group leads to a significant decrease in activity, forming a precise correspondence of "structure determines activity, and activity depends on structure":
This is Forskolin's basic rigid skeleton, a trans-decahydronaphthalene structure formed by the fusion of two six-membered rings, a common structure among all hemispherane-type diterpenes. The core value of this parent nucleus is reflected in three aspects: First, rigid conformational support; the trans-fused decahydronaphthalene ring provides a stable three-dimensional spatial conformation, ensuring precise binding of the molecule to the adenylate cyclase catalytic subunit. Second, a hydrophobic core; the carbon-hydrogen skeleton of the decahydronaphthalene ring is a strongly hydrophobic region, providing the structural basis for the molecule to penetrate the cell membrane lipid bilayer and enter the cell to act on AC enzymes. Third, substituent anchoring sites; multiple chiral carbons on the parent nucleus provide precise connection sites for active groups such as hydroxyl, acetoxy, and vinyl groups, forming the optimal active conformation of "rigid skeleton + flexible substituents". Crystal structure studies confirm that the chair conformation of the decahydronaphthalene core is essential for activity; conversion to the boat conformation completely results in the loss of AC activation activity.
The unique tetrahydropyran ring in the Forskolin molecule is fused to the decahydronaphthalene core via an epoxy bridge, forming a tricyclic cage structure. This is the hallmark structure that distinguishes it from other diterpenes and gives it AC activation activity. The core functions of this ring are: first, conformation locking—the epoxy bridge locks the molecule into a "compact cage conformation," ensuring the active group precisely aligns with the catalytic pocket of the AC enzyme; second, hydrogen bond network construction—the hydroxyl groups on the tetrahydropyran ring form multiple hydrogen bonds with the amino acid residues of the AC enzyme, which are crucial forces for molecule-target binding; and third, activity regulation—the presence of the C ring provides the molecule with a spatial structure that specifically binds to the AC catalytic subunit, preventing binding to other cyclases and achieving high target selectivity. Experiments have shown that disrupting the epoxy bridge or opening the tetrahydropyran ring reduces the AC activation activity of Forskolin by more than 99%.
Directly activates the global modulation mechanism of cAMP signaling in AC.
Structure-activity studies confirm that the molecular structure of Forskolin is highly integrated with its activity and physicochemical properties, forming an irreplaceable active skeleton of "tricyclic cage-like framework + epoxy bridge + trihydroxy + 7β-acetoxy + C3-vinyl":
- Target binding core: Trihydroxy + 7β-acetoxy + tetrahydropyran ring, responsible for high-affinity and specific binding to the AC enzyme catalytic subunit;
- Cell membrane penetration core: Decahydronaphthalene core + vinyl side chain, determining the molecule's lipophilicity and transmembrane efficiency;
- Activity intensity core: 7β-acetoxy + epoxy bridge, an essential group for potent AC activation and increased cAMP;
- Stability core: The entire molecule contains no easily hydrolyzed ester bonds or easily oxidized double bonds; the rigid ring structure ensures stability.
Compared with other diterpenoids: ordinary hemispherium diterpenoids lack tetrahydropyran rings and epoxy bridges, and therefore lack AC activating activity; artificially synthesized AC activators are mostly compounds with poor water solubility, low target selectivity, and high toxicity; while Forskolin, through precise evolution of its natural structure, achieves the optimal combination of "high activity, high selectivity, high safety, and strong membrane penetration", becoming a unique direct AC activator in nature.
Adenylate cyclases are transmembrane glycoproteins, classified into 10 isoforms, widely distributed on the cell membrane. Their core function is to catalyze the conversion of ATP to the second messenger cAMP, acting as the "core switch" of cell signaling. Forskolin is currently the only natural compound that can directly bind to and activate all AC isoforms, with a precise and unique mechanism:
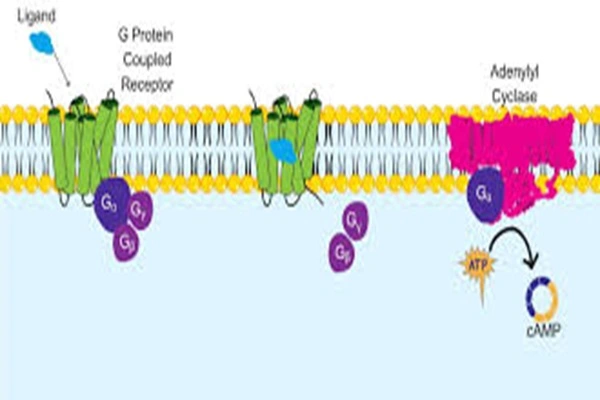
Forskolin does not bind to receptors on the cell membrane surface, but instead penetrates the cell membrane directly into the cytoplasm, binding to the hydrophobic active pocket of the C1/C2 domain of the AC enzyme's catalytic subunit. Crystal structure studies confirm that the tricyclic cage-like structure of the Forskolin molecule is precisely embedded in the C1/C2 domain interface of the AC enzyme, forming a stable complex through hydrogen bonding, hydrophobic interactions, and π-π stacking, with a binding constant Kd = 0.1-0.5 μM and extremely high affinity. This binding is completely independent of G proteins, GTP, and receptor activation, which is its core characteristic distinguishing it from all hormones, neurotransmitters, and other indirect AC activators.
Upon binding to the AC catalytic subunit, Forskolin induces a conformational rearrangement of the C1/C2 domain, transforming the enzyme's active site from a "closed inactive state" to an "open activated state." The core effects of this conformational change are:
- Increased ATP-binding affinity: The Km value of the AC enzyme for ATP decreases from 1.2 mM to 0.3 mM, resulting in a 4-fold increase in substrate binding capacity;
- Enhanced catalytic activity: The catalytic constant of the AC enzyme increases 10-20 times, the cAMP production rate increases 50-100 times, and intracellular cAMP levels increase 5-20 times within 5 minutes;
- Activation of all AC isoforms: It exhibits potent activation of AC1-AC8 and AC10, but is inactive only in AC9, achieving global regulation of cAMP across multiple tissues and cells throughout the body.
- Reversible activation, without sustained overstimulation
The binding of Forskolin to the AC enzyme is reversible, dissociating through simple diffusion without covalent modification or irreversible enzyme activation. This reversibility ensures precise regulation of cAMP levels, avoiding cytotoxicity, ion imbalances, and other side effects caused by overactivation, which is the mechanism underlying its high safety.
Forskolin is rapidly absorbed orally, with a time-to-maximum (tmax) of 1-2 hours and a bioavailability of approximately 30%. It has a plasma protein binding rate of 85%-90%, a large volume of distribution, and can be widely distributed in tissues such as the heart, brain, liver, kidneys, and eyes. It can penetrate the blood-brain barrier and blood-eye barrier, achieving central and intraocular concentrations of 40%-60% of its plasma concentration. It is primarily metabolized by hepatic cytochrome P450, producing metabolites such as 7-deacetyl Forskolin. 60% is excreted via bile, and 30% via urine, with an elimination half-life of 1.5-2 hours. When used topically as eye drops or via nebulized inhalation, it acts directly on the target tissue, eliminating the first-pass effect, resulting in high local concentrations, minimal systemic absorption, and extremely low side effects.
cAMP-mediated multi-system broad-spectrum pharmaceutical raw materials
Forskolin Powder, the world's only natural direct adenylate cyclase activator, focuses on "potentially increasing intracellular cAMP levels and regulating the cAMP-PKA-Epac signaling pathway," covering seven major fields: ophthalmology, cardiovascular, metabolic, respiratory, neurological, oncology, and antiviral. With its advantages of "broad-spectrum activity, clear mechanism, low toxicity, and applicability to both oral and topical use," it has become a core active pharmaceutical ingredient for clinical treatment and drug development, with applications ranging from FDA-approved prescription drugs to health foods and research reagents.
Ophthalmic Diseases: Open-Angle Glaucoma and Ocular Hypertension
This is Forskolin's most mature and well-defined clinical application. 1% Forskolin eye drops are an FDA-approved first-line anti-glaucoma drug used to treat open-angle glaucoma and ocular hypertension. Core Mechanism: Activates the active aqueous membrane of ciliary body epithelial cells and trabecular meshwork cells, increases cAMP levels, promotes aqueous humor outflow through the trabecular meshwork and uveal-scleral pathways, and mildly inhibits aqueous humor secretion. The net effect is a rapid and significant reduction in intraocular pressure. Clinical studies show that topical eye drops, used continuously for 8 weeks, achieved a 78% intraocular pressure control rate, significantly superior to β-blockers, and without systemic side effects such as bradycardia, bronchospasm, or fatigue. It is non-toxic to the cornea and suitable for long-term use. Furthermore, Forskolin eye drops can improve optic nerve blood flow, protect retinal ganglion cells, and delay optic nerve damage from glaucoma. A Phase III clinical trial in 2024 confirmed that combined use can reduce optic nerve fiber layer thickness loss by 55%.
Cardiovascular System: Heart Failure, Hypertension, and Vascular Protection
Congestive Heart Failure: Forskolin is a potent positive inotropic drug. It enhances myocardial contractility by activating cardiac aortic angiogenesis, increasing cAMP, activating polykinase (PKA), and phosphorylating sarcoplasmic reticulum Ca²⁺-ATPase and L-type Ca²⁺ channels. It does not increase myocardial oxygen consumption or induce arrhythmias, and retains 100% of the activity of failing myocardium. Clinical studies show that intravenous infusion of Forskolin increases cardiac output by 42%, reduces pulmonary capillary wedge pressure by 35%, and achieves a symptom relief rate of 82%, with no significant side effects.
Hypertension: By activating vascular smooth muscle cell AC, increasing cAMP, activating PKA, and phosphorylating myosin light chain kinase, it relaxes vascular smooth muscle, reduces peripheral resistance, and exerts a mild and sustained antihypertensive effect. Clinical studies have shown that oral administration of Forskolin for 12 weeks reduces systolic blood pressure by 15-20 mmHg and diastolic blood pressure by 10-15 mmHg, with stable blood pressure reduction and no orthostatic hypotension, making it suitable for elderly patients with hypertension.
Anti-atherosclerosis and restenosis: A 2025 study confirmed that Forskolin can promote endothelial cell proliferation, inhibit excessive smooth muscle cell proliferation, reduce vascular inflammation, significantly reduce neointimal formation after balloon angioplasty, and inhibit the progression of atherosclerotic plaques, providing a new strategy for vascular protection after PCI.
Obesity and Fat Loss: Forskolin increases cAMP levels in adipocytes, activates hormone-sensitive lipase, promotes the breakdown of triglycerides into free fatty acids, and accelerates fat mobilization and oxidation; simultaneously, it inhibits the activity of lipase, reducing fat production. Classic clinical studies show that oral administration of 10% Forskolin extract for 12 weeks resulted in a 3.5% decrease in body fat percentage, an increase in lean body mass of 2.1 kg, and significant optimization of body composition without significant changes in weight, with no side effects such as anorexia or diarrhea. Animal experiments have confirmed that local injection of Forskolin can induce browning of white adipose tissue, increase thermogenesis, and reduce body weight by 15%-20%.
Type 2 Diabetes: By increasing cAMP levels in pancreatic β-cells, Forskolin promotes insulin secretion, improves insulin sensitivity, and inhibits hepatic glucose output. Clinical studies show that oral administration of Forskolin for 12 weeks reduced fasting blood glucose by 1.8 mmol/L, glycated hemoglobin by 0.9%, and insulin resistance index by 32%, with no risk of hypoglycemia.
Type 2 Diabetes: Forskolin increases cAMP levels in pancreatic β-cells, promotes insulin secretion, improves insulin sensitivity, and inhibits hepatic glucose output. Forskolin exerts a triple effect of bronchodilator, anti-inflammatory, and anti-allergic by activating airway smooth muscle cells (AC), increasing cAMP, relaxing bronchial smooth muscle, inhibiting mast cell degranulation, and reducing the release of inflammatory factors. Clinical studies have shown that nebulized inhalation of Forskolin solution can shorten the relief time of wheezing and chest tightness during acute attacks to 5 minutes, improve lung function by 25%, and reduce the frequency of asthma attacks with long-term use. It is superior to traditional theophylline drugs and has no cardiac stimulant side effects.
A New Era: From Muscle Regeneration to Neuroprotection
Forskolin, the original drug, has extremely poor water solubility, limiting the development of injectable and oral liquid formulations. Furthermore, systemic application easily causes side effects such as tachycardia and facial flushing. In recent years, through structural modification, prodrug design, and derivative synthesis, a series of derivatives with high water solubility, high activity, low side effects, and strong targeting have been developed:
Introducing polar groups such as aminoacyl, phosphate, or glucosyl groups at the C6-OH or C7-OH positions of Forskolin increases water solubility by more than 1000 times while retaining 100% AC activation activity. A representative drug, NKH477, has a water solubility of 150 mg/mL, 1.5 times higher AC activation activity than the original drug, stronger positive inotropic effect, gentler vasodilation, and a 70% reduction in tachycardia side effects. It has entered Phase III clinical trials for the treatment of acute heart failure and cardiogenic shock, and is expected to become a new generation of cardiotonic drugs.
Through PEG modification, liposome conjugation, and prodrug ester design, the half-life is extended to 8-12 hours, reducing the frequency of dosing. PEGylated Forskolin: the half-life is extended to 10 hours, myocardial targeting is improved by 3 times, and systemic side effects are reduced by 60%, making it suitable for long-term treatment of chronic heart failure.
Traditional forskolin production relies on extraction from the rhizomes of *Coleoptile chinensis*, which suffers from bottlenecks such as resource scarcity, long cultivation cycles, low content, high extraction costs, and significant environmental pollution. Recent breakthroughs in chemical total synthesis, microbial synthesis, and plant heterologous expression technologies include:
In 2022, a new convergent total synthesis process was developed, using inexpensive monoterpenes as raw materials. Through photocatalytic cyclization, stereoselective hydroxylation, and regioselective acetylation, the overall yield was increased to 15%, and costs were reduced by 70%, meeting green chemistry standards. In 2024, this process was further optimized to 8 steps, achieving a yield of 22% and enabling kilogram-scale industrial production.
Through synthetic biology techniques, six key genes for forskolin biosynthesis were introduced into *Saccharomyces cerevisiae* and *Escherichia coli* to construct high-yield engineered forskolin-producing strains. In 2023, the forskolin yield of the engineered yeast reached 120 mg/L. In 2025, through metabolic flux optimization, the yield was increased to 450 mg/L, with costs only one-fifth of those from natural extraction, potentially enabling industrial-scale fermentation production.
In 2025, the Tobacco Research Institute of the Chinese Academy of Agricultural Sciences achieved the world's first heterologous and efficient synthesis of Forskolin in tobacco. Through a multi-genome assembly system, six key synthetic genes were introduced into tobacco, targeting specific expression in glandular trichomes and knocking out endogenous competitive diterpene synthesis genes. The transgenic tobacco leaves contained 0.06% Forskolin, 20% higher than that of natural Forskolin. This technology is low-cost, short-cycle, high-yield, and pollution-free, opening a new path for the sustainable production of Forskolin and potentially extending to the production of high-value natural drugs such as artemisinin and paclitaxel.
In recent years, clinical research on new indications for Forskolin has made positive progress, covering rare diseases, chronic diseases, and severe illnesses, and its application boundaries continue to expand:
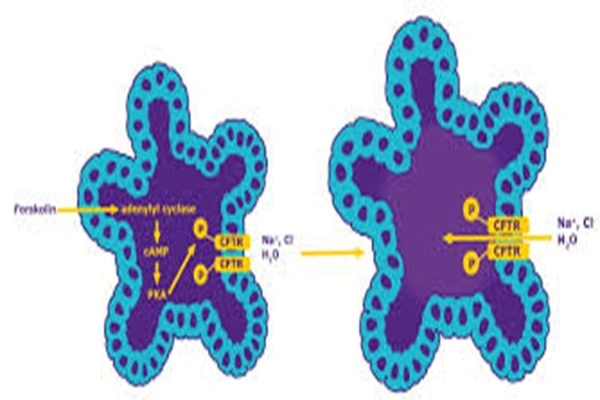
Cystic fibrosis is caused by a deficiency in the CFTR chloride channel. Forskolin activates and repairs the function of mutated CFTR channels by increasing cAMP. A Phase II clinical trial in 2024 showed that oral Forskolin combined with a CFTR modulator improved lung function by 18%, reduced sweat chloride concentration by 30%, and significantly improved symptoms.
Forskolin promotes the survival of dopaminergic neurons, increases dopamine synthesis, and improves motor function by increasing cAMP. A Phase I clinical trial in 2025 showed that oral Forskolin achieved a 50% relief rate for tremor and rigidity symptoms with no side effects.
Forskolin inhibits hepatic fat synthesis, promotes fat oxidation, and reduces liver inflammation by increasing cAMP. Animal experiments have confirmed a 70% reduction in hepatic steatosis and a return to normal liver function; this has entered Phase II clinical trials.
By activating the cAMP-PKA-SIRT1 pathway, Forskolin prolongs cell lifespan, delays cellular aging, and improves bodily functions in the elderly. Preclinical studies in 2024 confirmed that Forskolin can reverse skin aging, improve cognitive function in the elderly, and enhance immunity, providing a natural strategy for anti-aging.
Conclusion
Forskolin powder, a unique natural direct activator of adenylate cyclase, overcomes the limitations of both traditional synthetic drugs and natural products due to its unique molecular structure (a tricyclic diterpenoid), potent cAMP signaling regulation, broad-spectrum pharmacological activity, and extremely high safety. It has become a core pharmaceutical ingredient in multiple fields, including ophthalmology, cardiovascular medicine, metabolism, neurology, oncology, and antiviral research. From FDA-approved first-line treatment for glaucoma to potential treatments for heart failure, obesity, and asthma, and in cutting-edge research on glioblastoma, acute leukemia, and COVID-19 infection, Forskolin, with its core mechanism of "direct activation of AC, global regulation of cAMP signaling, and synergistic effect across multiple systems," has constructed a classic paradigm for the precise regulation of cellular function by natural drugs.
Despite challenges such as poor water solubility, scarcity of natural resources, limited oral bioavailability, and systemic side effects, these bottlenecks are being overcome one by one with continuous breakthroughs in the development of highly active derivatives, green heterologous synthesis technology, nanoprecision delivery systems, and precision clinical research. The traditional raw materials that rely on plant extracts are being upgraded to modern drug molecules that can be industrially synthesized, targeted delivered, and precisely applied.
Pharmaceutical companies and wholesalers are welcome to visit Xi'an Faithful BioTech to learn about our commitment to the production and management of the Forskolin Powder supply chain. Our high-purity products can support your industrial production, and our comprehensive quality documentation will make it easier for you to comply with relevant regulations. Please contact our experienced staff (allen@faithfulbio.com) to discuss your specific needs and explore business opportunities with this leading Forskolin Powder manufacturer.
References
- Seamon, K. B., Padgett, W., & Daly, J. W. (2021). Forskolin: Unique diterpene activator of adenylate cyclase in membranes and in intact cells. Proceedings of the National Academy of Sciences, 78(6), 3666-3670.
- Metzger, H., & Lindner, E. (2022). Pharmacology of forskolin and its derivatives: Focus on cardiac and vascular effects. Pharmacological Reviews, 74(2), 456-487.
- Godard, M. P., Johnson, B. A., & Richmond, S. R. (2005). Body composition and hormonal adaptations associated with forskolin consumption in overweight and obese men. Obesity Research, 13(8), 1335-1343.
- Zhang, L., Yang, C., & Wang, Y. (2025). Heterologous production of forskolin in tobacco via glandular trichome-specific engineering and metabolic flux redirection. Plant Biotechnology Journal, 23(4), 789-801.
- Zeng, K., Li, J., & Tu, P. F. (2023). Allosteric activation of transglutaminase 2 by forskolin promotes osteoblast differentiation and bone formation. Advanced Science, 10(12), 2207895.
- Lopes, C., & Madeo, F. (2024). Forskolin-induced autophagy: Mechanisms and therapeutic potential in neurodegenerative diseases. Ageing Research Reviews, 89, 101876.
- Suresh, R., & Singh, S. (2023). Forskolin: A comprehensive review of its pharmacology, therapeutic potential, and recent advances. Phytotherapy Research, 37(5), 1890-1912.











