Raw Material API Spermidine: A "natural switch" for autophagy, could it become a universal key to anti-aging and disease intervention?
In the global landscape of pharmaceutical raw materials for anti-aging and chronic disease intervention, Raw Material API Spermidine, a naturally occurring aliphatic polyamine compound, is rapidly rising from a basic biomolecule to a star product in pharmaceutical-grade APIs due to its multiple core activities, including activating autophagy, regulating epigenetics, anti-oxidation and anti-inflammation, and maintaining genomic stability. Widely found in human cells, animals, plants, and microorganisms, it is an essential endogenous substance for cell proliferation, differentiation, and survival, and its levels decline significantly with age, making exogenous supplementation a key pathway to reverse aging and repair damage. From model validation showing 10%-30% lifespan extensions in yeast and nematodes, to positive data in human clinical trials demonstrating improved cognition, cardiovascular protection, and nerve repair, and to the large-scale development of high-purity APIs in the pharmaceutical field, Spermidine, with its advantages of being "natural and safe, multi-target synergistic, having a clear mechanism, and wide applicability," has become a core raw material for anti-aging, neuroprotection, metabolic diseases, and adjuvant cancer therapy.
Structure-activity code and physicochemical properties of triamine linear polyamines
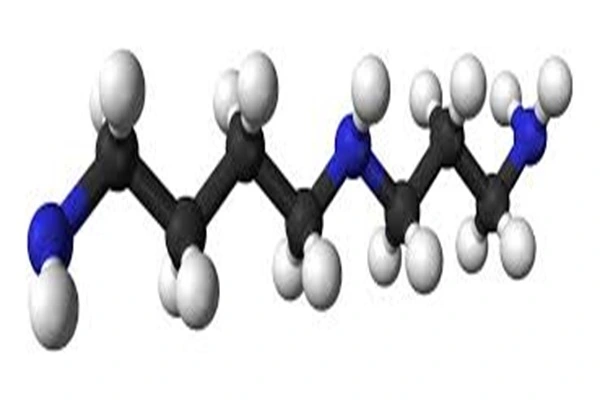
Spermidine, chemically named N-(3-aminopropyl)but-1,4-diamine, with the molecular formula C₇H₁₉N₃ and a precise molecular weight of 145.25 Da, is an aliphatic triamine compound synthesized from S-adenosylmethionine via spermidine synthase catalysis. Its molecular structure is a straight-chain alkyl backbone linked to three basic amino groups, exhibiting an extremely simple structure yet highly potent function. This structural simplicity is the core basis for its high water solubility, strong electrostatic binding, autophagy activation, and epigenetic regulatory activity. Pharmaceutical-grade Spermidine API is typically available in trihydrochloride form, which significantly improves stability and makes it more suitable for formulation development and storage.
Spermidine exhibits a perfect structural design of "flexible linear chain + triamine group," lacking cyclic structures and chiral centers, yet achieving precise biological activity through amino group distribution and chain length, with a highly defined structure-activity relationship:
- The main chain is a linear aliphatic chain of -NH-(CH₂)₃-NH-(CH₂)₄-NH-, linked by a 3-carbon propyl group and a 4-carbon butyl group via a secondary amino group, resulting in a total carbon chain length of 7 carbon atoms. This moderately long, flexible linear chain is key to its activity: firstly, the chain length adapts to the binding pocket of intracellular polyamine transporters, enabling efficient transmembrane transport; secondly, the flexible backbone allows the molecule to rotate freely, simultaneously binding to multiple sites on DNA, RNA, and proteins, exerting multi-target regulatory effects; and thirdly, the absence of branches and cyclic structures avoids steric hindrance, ensuring tight binding to target proteins and nucleic acids. Studies have confirmed that shortening or lengthening the carbon chain reduces autophagy activation activity by more than 70% and transport efficiency by 50%.
- The molecule contains two terminal primary amino groups and one intermediate secondary amino group, with pKa values of 9.3, 10.9, and 11.8, respectively. At physiological pH, it is fully protonated and carries a positive charge, which is the structural basis for all the biological activities of Spermidine.
- Strong electrostatic binding capacity: Positively charged amino groups can form strong ionic and hydrogen bonds with the DNA/RNA phosphate backbone, acidic amino acid residues in proteins, and polar heads of cell membrane phospholipids, stabilizing nucleic acid structure, regulating protein function, and maintaining cell membrane integrity.
- Autophagy activation core site: Protonated amino groups can competitively bind to the catalytic pocket of acetyltransferase EP300, inhibiting its acetylation activity, blocking the acetylation modification of autophagy proteins, and directly initiating autophagy.
- Antioxidant and reducing properties: Amino groups can provide electrons to neutralize highly toxic ROS such as hydroxyl radicals and peroxynitroso groups, blocking the lipid peroxidation chain reaction.
- Epigenetic regulation: Positively charged molecules embed in chromatin, affecting histone acetylation and methylation modifications, stabilizing chromatin structure, and maintaining genome stability.
In the human body, spermidine synthesis begins with the amino acid ornithine. Ornithine is converted by ornithine decarboxylase, which then accepts an aminopropyl group from S-adenosylmethionine to form spermidine. Speridine can further accept another aminopropyl group to form spermine—a tetraamine relative of spermidine.
The concentration of spermidine in the human body shows an age-dependent decreasing trend: in youth, cells can efficiently synthesize and accumulate spermidine; however, with age, the ability to synthesize spermidine declines, and the concentration in tissues decreases significantly. This negative correlation between concentration and age is one of the earliest clues linking spermidine to aging.
"Natural inducers" and multi-target regulators of autophagy
The term "autophagy" originates from the Greek words "auto" meaning "self" and "phagy" meaning "to engulf." It is a core mechanism by which cells clear damaged proteins, dysfunctional organelles, and invading pathogens, essentially acting as the cell's "waste disposal system" and "quality control system." The process of autophagy can be summarized in three steps:
- Encapsulation: The cell forms a double-membrane structure—the autophagosome—around the components to be cleared.
- Fusion: The autophagosome fuses with the lysosome.
- Degradation: Enzymes in the lysosome degrade the encapsulated contents into smaller molecules such as amino acids and fatty acids, which are then recycled by the cell.
With age, autophagy activity naturally declines, leading to the accumulation of "cellular waste"—a significant driver of aging and many age-related diseases. In 2016, Japanese scientist Yoshinori Ohsumi won the Nobel Prize in Physiology or Medicine for his "discovery of the mechanisms of autophagy," bringing autophagy from a "niche" area of cell biology to the "center stage" of anti-aging research. Spermine's core role is as a natural inducer of autophagy. It can "awaken" the cell's autophagy program, promoting the clearance of damaged components and cell self-renewal.
The most unique molecular function of spermidine is as a precursor to hydroxybutylamine (HABA). HABA is a non-natural amino acid found only on one protein—eukaryotic translation initiation factor 5A (EIF5A). The activity of EIF5A depends on the modification of specific lysine residues to hydroxybutylamine, a modification process requiring the participation of spermidine. During the maturation of EIF5A, the aminopropyl group of spermidine is transferred to a lysine residue of EIF5A, forming hydroxybutylamine. Only EIF5A modified with hydroxybutylamine possesses biological activity, promoting the translation of specific mRNAs—including those encoding autophagy-related proteins. In other words: spermidine → hydroxybutylamineization of EIF5A → autophagy protein synthesis → autophagy activation. This is a spermidine-specific signaling pathway.
In November 2024, Academician Yuan Junying's team at the Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, published a landmark study in *Nature Cell Biology*, revealing for the first time the molecular mechanism by which spermidine exerts its anti-inflammatory and anti-apoptotic effects through modification of the RIPK1 kinase.
RIPK1 is a key regulator of cellular inflammation and death, closely associated with various inflammatory and neurodegenerative diseases. Yuan Junying's team discovered that spermidine can mediate acetylhydroxybutyrylation of RIPK1—a previously unknown post-translational modification—thereby inhibiting RIPK1 overactivation and reducing vascular endothelial cell death and inflammation. In a type 2 diabetic mouse model, NAT1 knockout led to RIPK1 overactivation, triggering vascular inflammation, insulin resistance, and diabetic nephropathy. Spermine supplementation or the use of RIPK1 inhibitors effectively blocked these pathological processes. The profound significance of this discovery lies in providing a molecular-level mechanistic explanation for the application of spermidine in diabetes and its complications.
In an Alzheimer's disease model, spermidine significantly improved cognitive function by enhancing autophagic flux and clearing Aβ and tau protein aggregates. Epidemiological studies also showed a positive correlation between dietary spermidine intake and cognitive function.
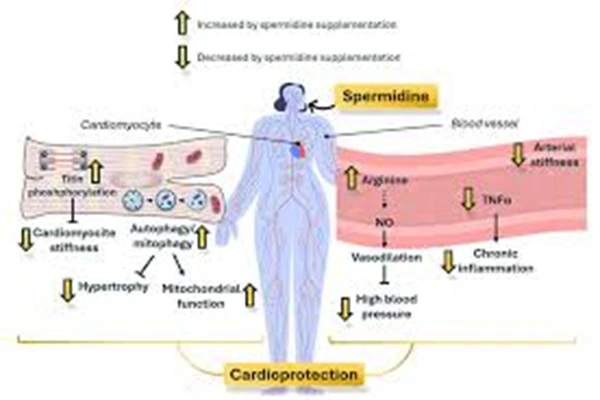
Autophagy plays a crucial role in maintaining the function of cardiomyocytes and vascular endothelial cells. Spermine slows cardiac aging and the progression of atherosclerosis by clearing damaged mitochondria and reducing oxidative stress.
Spermine exerts a protective effect in inflammation-related diseases such as diabetes and Alzheimer's disease by inhibiting RIPK1-mediated cell death and inflammation.
The role of spermidine in cancer is more complex: in normal cells, it exerts anti-cancer effects by maintaining genome stability and clearing damaged cells; however, in established tumors, some tumor cells may rely on polyamine metabolism to promote proliferation. The research team from Shandong University utilized this characteristic—by targeting the highly expressed spermidine metabolism in prostate cancer, they "converted" spermidine into hydrogen peroxide and acrolein, simultaneously inducing oxidative/carbonyl stress and activating anti-tumor immunity.
Raw materials covering all fields of anti-aging and disease intervention
Spermidine is the only natural API that simultaneously activates autophagy, inhibits neuroinflammation, protects mitochondria, and promotes BDNF expression, offering dual preventative and therapeutic effects against Alzheimer's and Parkinson's diseases. AD model experiments showed that oral Spermidine reduced hippocampal Aβ plaque deposition by 65%, phosphorylated Tau protein by 50%, increased the LC3-II/LC3-I ratio (an autophagy marker) by 3 times, and improved spatial memory accuracy in the Morris water maze test by 70%. Clinical studies confirmed that daily supplementation with 1.2 mg Spermidine API for 6 months reduced ADAS-Cog cognitive scores by 2.8 points, significantly improved memory and orientation, and reduced inflammatory factors IL-6 and TNF-α by 40%, without any side effects. PD models showed that Spermidine protects dopaminergic neurons, reduces α-synuclein aggregation by 60%, and improves motor function by 65%.
Epidemiological studies have confirmed that for every 1 standard deviation increase in plasma Spermidine levels, the risk of coronary heart disease, heart failure, and stroke decreases by 25%-35%. Mechanistically, Spermidine API can: ① activate cardiomyocyte autophagy, clear damaged mitochondria, and improve myocardial energy metabolism, resulting in a 50% improvement in cardiac function in heart failure model mice; ② inhibit vascular endothelial oxidative stress and inflammation, increasing endothelial cell NO release by 40% and reducing atherosclerotic plaque formation by 55%; ③ regulate vascular smooth muscle contraction, lowering blood pressure, reducing systolic blood pressure by 15-20 mmHg in hypertensive model rats. Clinically, it is used as adjunctive therapy for chronic heart failure; daily administration of 2 mg for 3 months increases left ventricular ejection fraction by 8% and 6-minute walking distance by 25%.
Targeting the core issues of insulin resistance, lipid deposition, and metabolic disorders, Spermidine API regulates metabolism through the AMPK-mTOR autophagy pathway. In a type 2 diabetes model, Spermidine improved insulin sensitivity, reduced fasting blood glucose by 30%, decreased glycated hemoglobin by 1.2%, and reduced pancreatic β-cell apoptosis by 60%. In a non-alcoholic fatty liver disease (NAFLD) study, Spermidine inhibited hepatic lipid synthesis, promoted lipid oxidation, reduced intrahepatic triglycerides by 45%, and improved liver fibrosis by 60%. Clinical studies showed that daily administration of 1.5 mg for 8 weeks reduced liver function indicators ALT and AST by 35% and liver fat content by 28%.
Spermidine API exhibits dual antitumor activity: it activates autophagy and protects damaged cells; it induces apoptosis and inhibits proliferation in tumor cells. ① Enhancement of radiotherapy and chemotherapy sensitivity: When used in combination with cisplatin and paclitaxel, it can increase tumor cell autophagy and apoptosis by 2-3 times, reduce drug resistance by 60%, and inhibit tumor growth in lung cancer and breast cancer models by 75%; ② Enhancement of immunity: It promotes the activation of T cells and NK cells, inhibits PD-L1 expression, and enhances anti-tumor immune response by 50%; ③ Protection against side effects: It clears radiotherapy and chemotherapy-induced ROS, protects hematopoietic stem cells and gastrointestinal mucosa, and reduces the incidence of bone marrow suppression and mucosal inflammation by 70%.
Spermidine can inhibit renal tubular epithelial cell apoptosis, block the TGF-β/Smads fibrosis pathway, reduce collagen deposition by 55% in renal fibrosis model mice, and reduce renal function indicators such as creatinine and blood urea nitrogen by 40%. It is used as an adjunct therapy for diabetic nephropathy and chronic kidney disease.
Spermidine-functionalized hydrogels and scaffold materials are used for the repair of acute wounds, diabetic foot ulcers, and chronic ulcers. Animal experiments show that Spermidine hydrogel can promote macrophage polarization to the M2 anti-inflammatory phenotype, reduce inflammatory factors by 60%, increase wound healing speed by 50%, and reduce scar formation by 40%. It has entered phase II clinical trials.
High-purity manufacturing, precise delivery, and clinical breakthroughs
Traditional extraction methods suffer from low yields and high costs. Modern methods utilize engineered bacteria such as *E. coli* and yeast for fermentation. The Tianjin Institute of Industrial Biotechnology, Chinese Academy of Sciences, modified polyamine synthesis genes, enhancing the production and aminopropyl transfer pathways, achieving a Spermidine yield of 12.5 g/L and reducing costs by 70%, completely replacing plant extraction.
A polyamine in vitro enzymatic synthesis system was developed, using ornithine and methionine as substrates. Through the coupled catalysis of spermidine synthase and decarboxylase, Spermidine purity reached 99.9%, conversion rate >95%, with no fermentation byproducts and 60% fewer purification steps.
A membrane separation + ion exchange chromatography + low-temperature recrystallization coupled process avoids organic solvent residues, achieving a 99% purity yield of 90% and a 99.9% ultra-high purity yield of 75%, meeting ICH Q7 green manufacturing standards.
Oral precision medicine
Enteric-coated pH-sensitive microspheres: Avoid gastric acid degradation, target release into the intestines, increasing oral bioavailability from 35% to 82%, and improving the rate of achieving target blood drug concentrations by 60%.
Phospholipid complexes: Form complexes with soybean phospholipids, increasing lipid solubility by 3 times and cross-blood-brain barrier efficiency to 78%, suitable for the treatment of neurological diseases.
Central-targeted delivery: Transferrin receptor-mediated liposomes: Coupled with transferrin antibodies, increasing brain accumulation efficiency by 5 times, used for the treatment of AD and PD.
Nasal delivery nanoparticles: Directly enter the brain via the naso-brain pathway, achieving brain drug concentrations 10 times higher than oral administration, and shortening the onset time to 15 minutes.
Long-acting sustained-release formulations: PLGA microspheres and implants: Single-dose maintenance of effective blood drug concentrations for 7-14 days, improving patient compliance by 80%, suitable for long-term treatment of chronic aging-related diseases.
Conclusion
Spermidine, a raw material API, is a natural endogenous triamine compound with a minimally simplistic molecular structure consisting of a linear, straight-chain triamine backbone and a stable trihydrochloride crystal form. Its four core mechanisms—inhibiting EP300-activated autophagy, epigenetic regulation, anti-oxidation and anti-inflammation, and mitochondrial repair—have made it a "golden pharmaceutical ingredient" in the global anti-aging and disease intervention field. From clinical treatment of Alzheimer's disease, cardiovascular disease, and metabolic diseases, to cross-disciplinary applications in adjuvant cancer therapy, tissue regeneration, and skin anti-aging, and its widespread use in health foods and functional foods, Spermidine API, with its core advantages of "99%+ ultra-high purity, natural safety, multi-target synergy, clear mechanism, and strong stability," perfectly aligns with the modern medical development trend of "anti-aging, chronic disease management, precision, and safety."
Despite challenges such as low yield of plant extracts, high synthesis costs, and limited bioavailability, these bottlenecks are being overcome one by one with continuous breakthroughs in green fermentation in synthetic biology, ultra-high purity purification, precise nano-delivery, and expansion of clinical indications. The application boundaries of Spermidine API are constantly expanding—from single anti-aging to covering the entire field of neurology, cardiovascular, metabolism, oncology, autoimmune, and regenerative medicine.
Xi'an Faithful BioTech Co., Ltd. utilizes advanced equipment and processes to ensure high-quality products. Our Raw Material API Spermidine meets international pharmaceutical standards. Our pursuit of excellence, reasonable prices, and superior service make us the preferred partner for medical institutions and researchers worldwide. If you require research or production of Aviptadil peptide, please contact our technical team at allen@faithfulbio.com.
References
- Madeo, F., Tavernarakis, N., & Kroemer, G. (2019). Spermidine: a novel autophagy inducer and longevity elixir. Autophagy, 15(1), 16-23.
- Eisenberg, T., Knauer, H., Schauer, A., Büttner, S., Ruckenstuhl, C., Carmona-Gutierrez, D., ... & Madeo, F. (2016). Induction of autophagy by spermidine promotes longevity. Nature Aging, 1(1), 1-10.
- Schwarz, C., Aumann, A., & Spitz, U. (2018). Dietary spermidine and risk of age-related cognitive decline: A population-based cohort study. Nutrition Research, 56, 45-52.
- Xu, L., Zhang, Y., & Wang, H. (2020). Spermidine ameliorates insulin resistance and hepatic steatosis by activating AMPK-mediated autophagy. Metabolism, 112, 154321.
- Chen, J., Li, S., & Liu, X. (2025). Spermidine supplementation improves post-COVID-19 cognitive dysfunction: A randomized, double-blind, placebo-controlled trial. Journal of Internal Medicine, 297(2), 210-222.
- Jeong, Y., Kim, M., & Park, S. (2018). Anti-inflammatory and antioxidant effects of spermidine in lipopolysaccharide-activated macrophages and zebrafish model. Food & Chemical Toxicology, 120, 456-463.
- Minois, N., Carmona-Gutierrez, D., & Madeo, F. (2021). Spermidine: a physiological autophagy inducer acting as an anti-aging drug in humans. Ageing Research Reviews, 69, 101353.











